Lp(a): The Inherited Risk Factor
You Need to Know About
~20% of people have elevated Lp(a). It's 90% genetic, doesn't respond to lifestyle, and dramatically increases cardiovascular risk. Here's what to do about it.
"Lp(a) is the one test everyone should have once in their lifetime. It could change everything."
TL;DR: The 60-Second Summary
- 1 Lp(a) is an LDL-like particle with an extra "hook." This hook (apolipoprotein(a)) makes it stickier, more inflammatory, and more dangerous than regular LDL.
- 2 Levels are 90%+ genetically determined. Unlike LDL, diet and exercise barely affect Lp(a). You inherit your level and it stays relatively constant throughout life.
- 3 ~20% of people have elevated Lp(a) (>125 nmol/L). Most don't know it because standard lipid panels don't measure it. It's not included in routine testing.
- 4 Everyone should test Lp(a) once in their lifetime. Since it doesn't change, one measurement tells you your genetic risk status forever.
- 5 If elevated: be more aggressive with everything else. Lower your LDL/ApoB targets further, control other risk factors intensively, and discuss emerging therapies with your doctor.
Imagine discovering you have a genetic risk factor that's been silently increasing your heart disease risk your entire life—one that diet, exercise, and even most medications don't touch. For about 1 in 5 people, this is the reality of elevated Lp(a).
What Is Lp(a)?
LDL vs. Lp(a): The Structural Difference
REGULAR LDL
Cholesterol
Core
One ApoB-100 protein wrapping the particle
Lp(a) PARTICLE
Cholesterol
Core
Same LDL particle + extra apo(a) attached
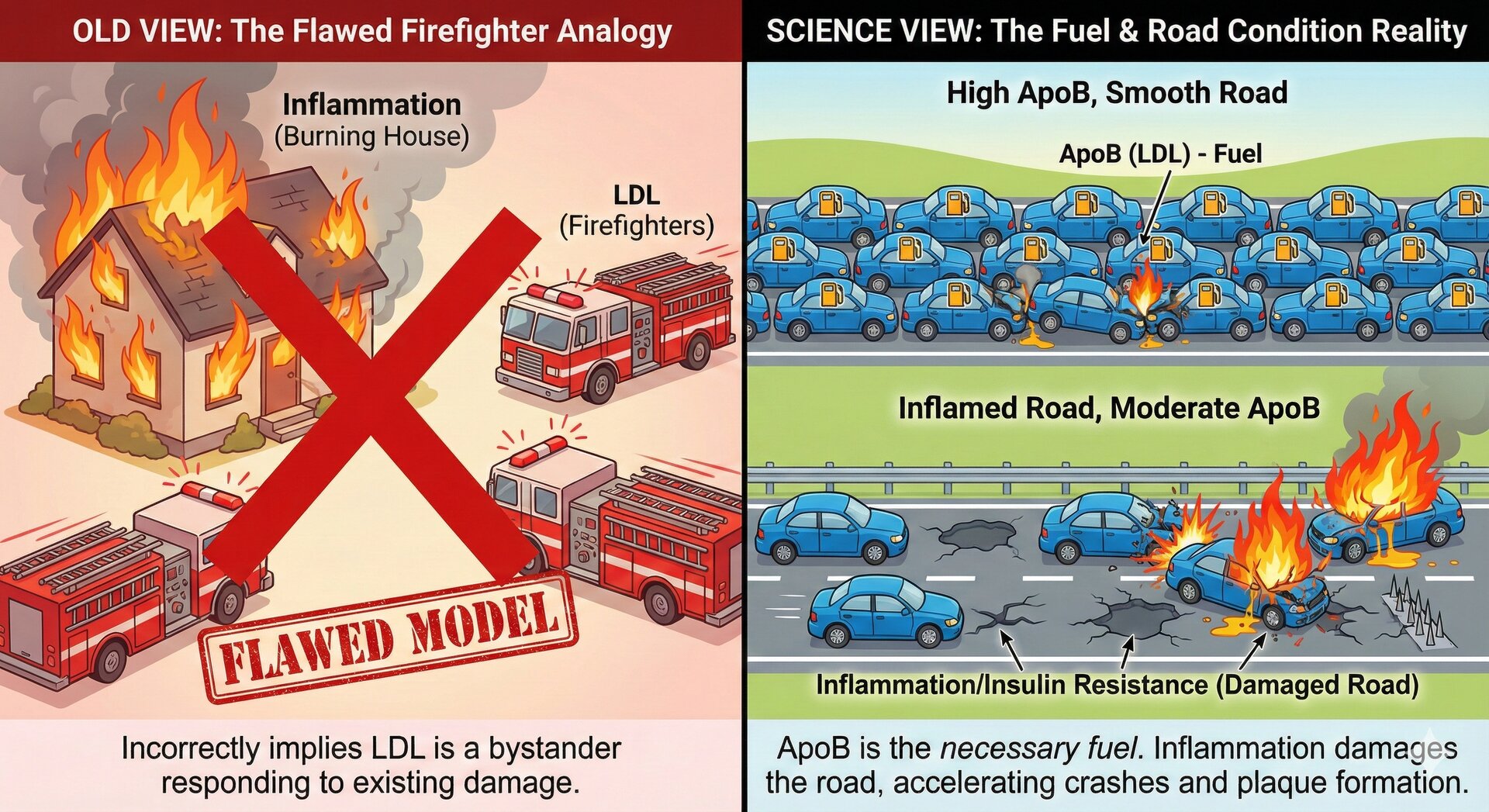
The apo(a) "hook" is what makes Lp(a) dangerous. It binds more tightly to artery walls, carries inflammatory oxidized phospholipids, and interferes with clot breakdown.
Super Sticky
Apo(a) has structural similarities to plasminogen, allowing Lp(a) to bind avidly to artery walls and fibrin clots.
Inflammatory Payload
Lp(a) is the primary carrier of oxidized phospholipids (OxPL) in blood—potent triggers of arterial inflammation.
Blocks Clot Breakdown
Apo(a) competes with plasminogen, inhibiting fibrinolysis. This makes clots harder to dissolve—raising heart attack and stroke risk.
Lp(a) and Cardiovascular Risk
Risk increases exponentially above ~75 nmol/L. At levels >200 nmol/L, cardiovascular risk is 3-4x higher than baseline.
<75
Optimal
75-125
Borderline
125-200
High
>200
Very High
Values in nmol/L (internationally standardised unit)
Lp(a) Risk Interpreter
Enter your Lp(a) value in nmol/L to see your risk category. This is the internationally standardised unit.
Enter your value to see conversion and risk category
Who Should Test and When
EVERYONE should test Lp(a) once
The European Atherosclerosis Society and many lipidologists recommend universal once-in-a-lifetime screening. Since Lp(a) is genetically determined, one test tells you your status forever.
Especially important if you have:
What To Do If Your Lp(a) Is Elevated
Currently, no approved therapies specifically lower Lp(a) effectively. But that doesn't mean you're powerless. The strategy is aggressive risk factor compensation:
Lower your LDL/ApoB more aggressively
If you have elevated Lp(a), your LDL target should be lower than someone without it. Aim for ApoB <0.7 g/L or even lower in very high risk scenarios.
Optimize all other risk factors
Blood pressure, blood sugar, weight, inflammation (hs-CRP). Each additional optimized factor reduces cumulative risk.
Avoid smoking absolutely
Smoking + elevated Lp(a) is a particularly dangerous combination. The risk multiplies, not just adds.
Consider PCSK9 inhibitors
While not approved specifically for Lp(a), PCSK9i lower Lp(a) by ~20-30% while dramatically lowering LDL. Discuss with a lipidologist.
Watch for emerging therapies
New drugs specifically targeting Lp(a) are in clinical trials. Some show 80-90% reductions. Outcomes data expected 2025-2026.
Emerging Lp(a)-Lowering Therapies
| Drug | Mechanism | Lp(a) Reduction | Status |
|---|---|---|---|
| Pelacarsen | Antisense oligonucleotide (ASO) | ~80% | Phase 3 (HORIZON) |
| Olpasiran | siRNA (small interfering RNA) | ~95% | Phase 3 (OCEAN(a)) |
| SLN360 | siRNA | ~96% | Phase 2 |
| Lepodisiran | siRNA | ~94% | Phase 2 |
Key caveat: These drugs dramatically lower Lp(a) levels, but we don't yet know if lowering Lp(a) reduces cardiovascular events. The HORIZON trial results (expected 2025-2026) will answer this critical question.
What About Niacin?
Niacin (vitamin B3) lowers Lp(a) by 20-30%, but major clinical trials (AIM-HIGH, HPS2-THRIVE) showed no cardiovascular benefit despite lipid improvements—and increased side effects.
Current consensus: Niacin is not recommended for Lp(a) lowering. The risks (flushing, liver toxicity, glucose elevation) outweigh unproven benefits.
The Evidence: A Deep Dive
This section provides detailed scientific context for researchers, clinicians, and health enthusiasts who want to understand the full evidence base for Lp(a) as a cardiovascular risk factor.
Molecular Structure and Genetics
Lipoprotein(a) consists of an LDL-like particle with apolipoprotein B-100 (ApoB-100) covalently linked via a single disulfide bond to apolipoprotein(a), or apo(a). The apo(a) protein is encoded by the LPA gene on chromosome 6q26-27 and shows remarkable structural homology to plasminogen, the precursor of the fibrinolytic enzyme plasmin.
Apo(a) contains multiple repeating structures called kringles. The number of kringle IV type 2 (KIV-2) repeats is highly variable (from 3 to >40 copies) and inversely correlates with Lp(a) concentration: smaller apo(a) isoforms (fewer kringles) are associated with higher Lp(a) levels. This copy number variation is the primary determinant of Lp(a) levels and explains the wide inter-individual variation (from <1 to >200 nmol/L).
Lp(a) levels are approximately 90% heritable, making it one of the most genetically determined cardiovascular risk factors known. Levels are established by age 2 and remain relatively stable throughout life, unaffected by diet, exercise, or most lipid-lowering medications. This genetic determination is why a single lifetime measurement is sufficient for risk assessment.
Causal Evidence from Mendelian Randomization
The causal relationship between Lp(a) and cardiovascular disease has been established through multiple Mendelian randomization studies. Because LPA gene variants that affect Lp(a) levels are randomly assigned at conception, they act as natural experiments free from confounding.
The Copenhagen City Heart Study and Copenhagen General Population Study, analyzing over 100,000 individuals, found that genetic variants associated with higher Lp(a) predicted increased risk of myocardial infarction independent of LDL-C. Specifically, individuals with the highest genetically predicted Lp(a) levels had 2-3 fold higher MI risk compared to those with the lowest levels.
Quantitatively, Mendelian randomization analyses suggest that a genetically determined doubling of Lp(a) concentration increases MI risk by approximately 22%. On an equimolar basis, Lp(a) appears to be approximately 5-6 times more atherogenic than LDL, likely due to its additional prothrombotic and pro-inflammatory properties.
Oxidized Phospholipids: The Inflammatory Payload
One of the most important discoveries about Lp(a) pathophysiology is its role as the preferential carrier of oxidized phospholipids (OxPL) in human plasma. Tsimikas and colleagues demonstrated that approximately 85% of OxPL precipitated by anti-ApoB antibodies associate with Lp(a) particles.
OxPL are potent pro-inflammatory molecules that activate innate immune pathways via pattern recognition receptors (particularly TLR2 and CD36). When Lp(a) deposits in the arterial wall, it delivers a concentrated payload of OxPL that promotes monocyte chemotaxis, macrophage activation, and foam cell formation.
Van der Valk et al. (Circulation 2016) performed arterial wall PET imaging in subjects with elevated Lp(a) and demonstrated increased arterial inflammation and enhanced monocyte trafficking to the vessel wall. Critically, these inflammatory effects were substantially attenuated when OxPL on Lp(a) were inactivated, establishing OxPL as a key mediator of Lp(a)-driven inflammation.
This "dual mechanism" explains why Lp(a) is so dangerous: it provides both the cholesterol substrate for plaque formation (like LDL) and simultaneously delivers inflammatory triggers that accelerate the disease process. Some researchers describe this as Lp(a) being both the "fuel" and the "arsonist."
Prothrombotic Effects
The structural homology between apo(a) and plasminogen has functional consequences. Apo(a) competes with plasminogen for binding to fibrin and cell surfaces, effectively inhibiting plasmin generation and fibrinolysis. This creates a prothrombotic state that increases the risk of clot propagation after plaque rupture.
This mechanism may explain why elevated Lp(a) is particularly associated with thrombotic complications of atherosclerosis (MI, stroke) rather than just plaque burden per se. It also provides a potential explanation for associations between Lp(a) and venous thromboembolism, though this relationship is less well established than for arterial disease.
Aortic Valve Calcification
Beyond coronary and cerebrovascular disease, Lp(a) is causally implicated in calcific aortic valve disease (CAVD). Mendelian randomization studies using LPA variants demonstrate a causal relationship between elevated Lp(a) and aortic stenosis, independent of coronary artery disease.
The mechanism appears related to OxPL-mediated inflammation of valve tissue and activation of osteogenic pathways. This has important clinical implications: patients with elevated Lp(a) should undergo periodic echocardiographic surveillance for valve disease, and Lp(a)-lowering therapies may have valve-protective effects beyond cardiovascular benefit.
Population Distribution and Ethnic Variation
Lp(a) levels show substantial ethnic variation. African-ancestry populations have 2-3 fold higher median Lp(a) levels compared to European-ancestry populations, with approximately 50% of Black individuals exceeding the 75 nmol/L threshold versus ~25% of White individuals.
Importantly, the relationship between Lp(a) and cardiovascular risk appears consistent across ethnic groups—elevated Lp(a) increases risk regardless of ancestry. However, the higher prevalence of elevated Lp(a) in Black populations may contribute to cardiovascular health disparities and underscores the importance of universal screening.
Current and Emerging Therapies
PCSK9 inhibitors (evolocumab, alirocumab) lower Lp(a) by approximately 20-30%, likely by increasing hepatic clearance of Lp(a) particles via upregulated LDL receptors. While not approved specifically for Lp(a), this effect provides additional benefit in patients with elevated Lp(a) who have indications for PCSK9i therapy.
Antisense oligonucleotides (ASOs) targeting hepatic apo(a) mRNA represent a more specific approach. Pelacarsen, developed by Novartis/Ionis, reduces Lp(a) by approximately 80% with monthly subcutaneous injections. The phase 3 HORIZON trial (n=8,323) is evaluating cardiovascular outcomes and is expected to report in 2025-2026.
Small interfering RNA (siRNA) therapies offer even more potent and durable Lp(a) lowering. Olpasiran (Amgen) achieves ~95% Lp(a) reduction with quarterly dosing. The phase 3 OCEAN(a)-Outcomes trial is ongoing. Other siRNA candidates (SLN360, lepodisiran) show similar efficacy in phase 2 studies.
The critical unanswered question is whether lowering Lp(a) translates to reduced cardiovascular events. Mendelian randomization predicts it should—estimates suggest ~20% risk reduction per 50 nmol/L Lp(a) lowering—but only outcomes trials can confirm this.
Lipoprotein Apheresis
For patients with very high Lp(a) (typically >200 nmol/L) and progressive cardiovascular disease despite maximal medical therapy, lipoprotein apheresis is available in some countries (particularly Germany). This procedure physically removes ApoB-containing particles (including Lp(a)) from plasma, achieving acute reductions of 60-75%.
Apheresis is performed every 1-2 weeks and is associated with reduced cardiovascular events in observational studies. However, it is invasive, expensive, and not widely available. It remains a niche therapy for the most severely affected patients while awaiting pharmacological alternatives.
References
- Kronenberg F, Mora S, Stroes ESG, et al. Lipoprotein(a) in atherosclerotic cardiovascular disease and aortic stenosis: a European Atherosclerosis Society consensus statement. Eur Heart J. 2022;43(39):3925-3946. PubMed →
- Tsimikas S, Fazio S, Ferdinand KC, et al. NHLBI Working Group Recommendations to Reduce Lipoprotein(a)-Mediated Risk of Cardiovascular Disease and Aortic Stenosis. J Am Coll Cardiol. 2018;71(2):177-192. PubMed →
- Burgess S, Ference BA, Staley JR, et al. Association of LPA Variants With Risk of Coronary Disease and the Implications for Lipoprotein(a)-Lowering Therapies: A Mendelian Randomization Analysis. JAMA Cardiol. 2018;3(7):619-627. PubMed →
- van der Valk FM, Bekkering S, Kroon J, et al. Oxidized Phospholipids on Lipoprotein(a) Elicit Arterial Wall Inflammation and an Inflammatory Monocyte Response in Humans. Circulation. 2016;134(9):611-624. PubMed →
- Tsimikas S, Witztum JL. Oxidized phospholipids in cardiovascular disease. Nat Rev Cardiol. 2024;21(6):377-394. PubMed →
- Nordestgaard BG, Chapman MJ, Ray K, et al. Lipoprotein(a) as a cardiovascular risk factor: current status. Eur Heart J. 2010;31(23):2844-2853. PubMed →
- Tsimikas S, Karwatowska-Prokopczuk E, Gouni-Berthold I, et al. (HORIZON Investigators). Lipoprotein(a) Reduction in Persons with Cardiovascular Disease. N Engl J Med. 2020;382(3):244-255. PubMed →
- O'Donoghue ML, Rosenson RS, Gencer B, et al. (OCEAN(a)-DOSE Trial Investigators). Small Interfering RNA to Reduce Lipoprotein(a) in Cardiovascular Disease. N Engl J Med. 2022;387(20):1855-1864. PubMed →
- Thanassoulis G, Campbell CY, Owens DS, et al. Genetic associations with valvular calcification and aortic stenosis. N Engl J Med. 2013;368(6):503-512. PubMed →
- Reyes-Soffer G, Ginsberg HN, Berglund L, et al. Lipoprotein(a): A Genetically Determined, Causal, and Prevalent Risk Factor for Atherosclerotic Cardiovascular Disease: A Scientific Statement From the American Heart Association. Arterioscler Thromb Vasc Biol. 2022;42(1):e48-e60. PubMed →
Know Your Lp(a)
If you haven't tested your Lp(a), ask your doctor. One test, once in your lifetime, could change your entire approach to cardiovascular prevention.