Lowering Cholesterol:
Making the Right Choice
When lifestyle is enough, when medication makes sense, and how to match treatment intensity to your actual risk
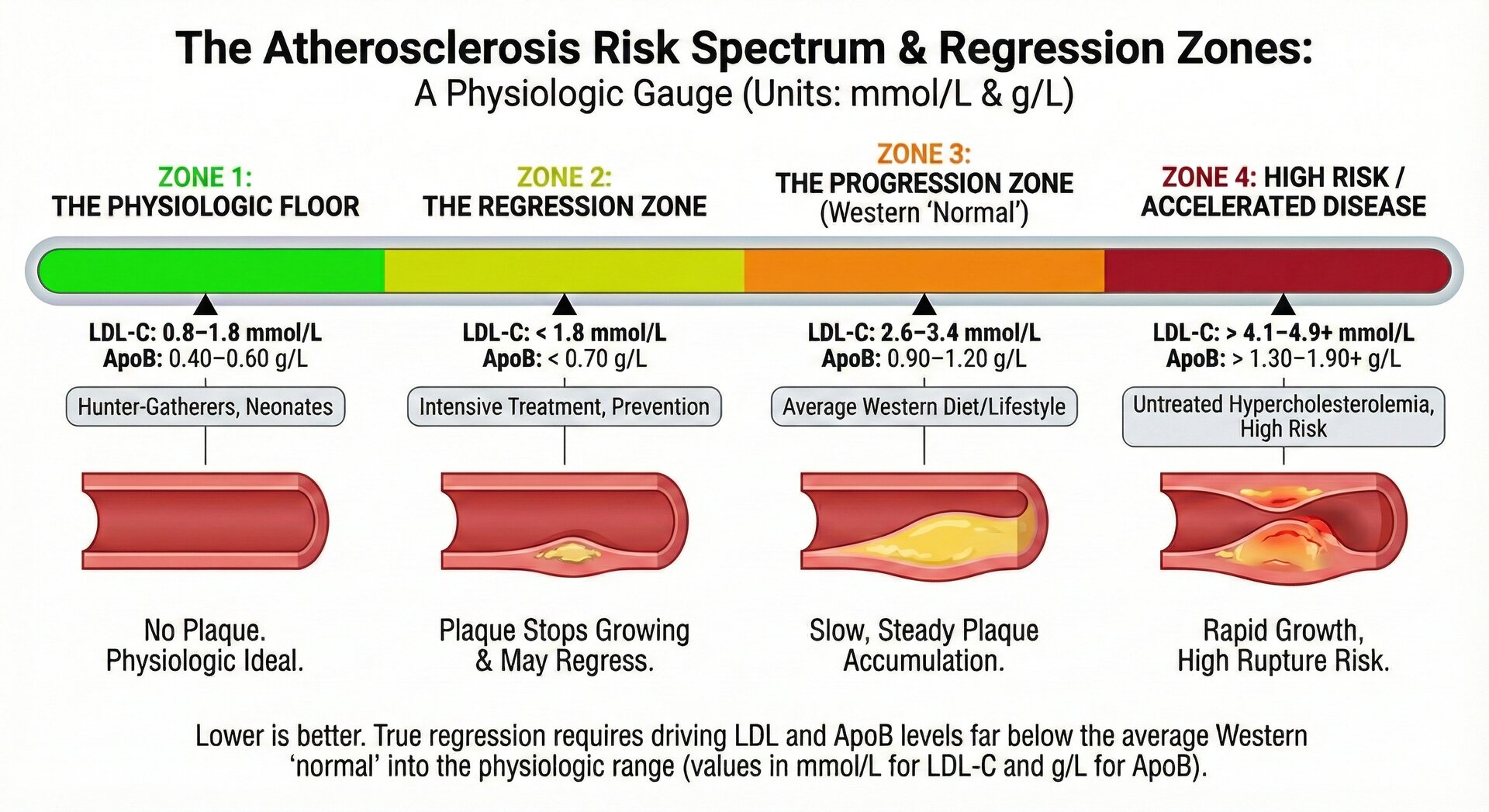
"Match the treatment to the risk. Aggressive intervention for high risk, patience and lifestyle for low risk."
TL;DR: The 60-Second Summary
- 1 Lifestyle works—but has limits. Diet, exercise, and weight loss can lower LDL by 10-30%. For some, that's enough. For others, it barely scratches the surface.
- 2 Statins remain first-line for most who need medication. They're safe, effective, and cheap. Most "side effects" are nocebo (caused by expectation, not the drug).
- 3 PCSK9 inhibitors are powerful but expensive. They lower LDL by ~60% and are reserved for high-risk patients who can't reach targets with statins alone.
- 4 Match intensity to risk level. CAC = 0 and low risk? Lifestyle focus. CAC > 100 or existing disease? Aggressive pharmacotherapy.
- 5 The goal isn't "lowering numbers"—it's preventing events. Whether you need medication depends on your total cardiovascular risk, not just a single LDL value.
The decision to start cholesterol medication is deeply personal. It depends on your risk profile, values, and goals. Here's a framework to help you and your doctor make the right choice for your situation.
Lifestyle Interventions
How Much Can Lifestyle Lower LDL?
Effect sizes vary by individual. Some people are "hyper-responders" to diet; others see minimal change.
Diet Changes
- Reduce saturated fat → 5-10% LDL reduction
- Add soluble fiber (oats, psyllium) → 5-10%
- Plant sterols/stanols → 5-15%
- Mediterranean pattern → 10-15% (plus other benefits)
Exercise & Weight
- Aerobic exercise → Modest LDL effect, major HDL/TG benefit
- Weight loss (10%) → 5-8% LDL reduction
- Visceral fat loss → Improves particle profile
- Combined effect → Up to 20-30% in best responders
The limitation: For someone with LDL of 4.7 mmol/L needing to reach <1.8 mmol/L (very high risk target), lifestyle alone rarely gets there. A 20% reduction brings you to 3.7 mmol/L—still far from goal. This is when medication becomes necessary.
Medication Options
| Drug Class | LDL Reduction | Cost | Key Notes |
|---|---|---|---|
| Statins | 30-55% | $ | First-line for most patients. Generic, well-studied. |
| Ezetimibe | 15-20% | $ | Blocks cholesterol absorption. Good add-on to statins. |
| PCSK9 Inhibitors | 50-65% | $$$ | Injectable. Reserved for high risk or statin-intolerant. |
| Inclisiran | 50% | $$$ | siRNA, twice-yearly injection. Long-acting PCSK9 inhibition. |
| Bempedoic Acid | 15-25% | $$ | Oral, for statin-intolerant. Doesn't cause muscle symptoms. |
Statins: Addressing the Concerns
Common Concerns
"Statins cause muscle pain"
Real myalgia occurs in ~5% of patients. But the SAMSON trial showed when patients didn't know if they were taking statin or placebo, symptom rates were nearly identical. Most "statin intolerance" is nocebo effect.
"Statins cause diabetes"
Slightly accelerate diabetes diagnosis in those already predisposed (~1 extra case per 1,000 treated per year). But cardiovascular benefit far outweighs this risk.
"Statins cause memory problems"
Large meta-analyses and the HOPE-3 trial show no cognitive decline. Anecdotal reports exist but aren't supported by controlled data.
The SAMSON Trial: Proving Nocebo
Patients cycled through months of: statin, placebo, or nothing. They reported symptoms daily.
90% of symptoms attributed to statins occurred equally with placebo. Taking any pill caused symptoms—not the statin specifically.
Risk-Based Decision Framework
Match Treatment Intensity to Risk Level
CAC = 0, No diabetes, 10-year risk <5%
Approach: Lifestyle optimization. Recheck in 5-10 years.
Medication rarely needed unless FH, very high LDL (>190), or elevated Lp(a).
CAC 1-99, or 10-year risk 5-10%
Approach: Aggressive lifestyle. Consider moderate-intensity statin.
Target LDL <2.6 mmol/L or ApoB <0.9 g/L. Shared decision-making.
CAC 100-399, Diabetes, or 10-year risk 10-20%
Approach: High-intensity statin. Add ezetimibe if needed.
Target LDL <1.8 mmol/L or ApoB <0.8 g/L. Lifestyle + medication.
CAC >400, Existing ASCVD, or Multiple high-risk conditions
Approach: Maximal tolerated statin + ezetimibe. Consider PCSK9i.
Target LDL <1.4 mmol/L or ApoB <0.65 g/L. Aggressive, guideline-directed therapy.
Guideline-Based Targets
| Risk Category | LDL-C Target | ApoB Target |
|---|---|---|
| Low Risk | <3.0 mmol/L | <1.0 g/L |
| Moderate Risk | <2.6 mmol/L | <0.9 g/L |
| High Risk | <1.8 mmol/L | <0.8 g/L |
| Very High Risk / ASCVD | <1.4 mmol/L | <0.65 g/L |
Based on 2019 ESC/EAS Guidelines. ACC/AHA guidelines use similar thresholds. Always individualize based on total risk profile.
Monitoring Your Treatment
After starting medication
Recheck lipids at 4-12 weeks. This confirms response and allows dose adjustment.
Once stable
Annual lipid panel is typically sufficient. More frequent if adjusting therapy.
Liver function
Check baseline ALT before starting. Routine monitoring not needed unless symptoms.
Muscle symptoms
Report significant new muscle pain. CK testing only if symptoms are severe.
The Evidence: A Deep Dive
This section provides detailed scientific evidence for treatment recommendations, including major trial data and guideline development rationale.
Statin Evidence Base
Statins are the most extensively studied cardiovascular drugs in history. The Cholesterol Treatment Trialists' (CTT) Collaboration meta-analyses, incorporating individual patient data from over 170,000 participants across 27 randomized trials, established the foundation for modern statin guidelines.
Key findings from CTT meta-analyses:
- Each 1 mmol/L reduction in LDL-C reduces major vascular events by approximately 22%
- This benefit is consistent regardless of baseline LDL-C level—even those starting below 2.6 mmol/L benefit
- Benefit accrues over time: year 1 shows ~12% reduction, but years 2+ show the full ~22% per mmol/L
- The relationship is log-linear with no evidence of a threshold below which benefit stops
Absolute benefit depends on baseline risk: A 22% relative reduction means different things for different people. For someone with 2% 10-year risk, statins prevent ~4 events per 1,000 treated over 10 years. For someone with 30% risk, that same relative reduction prevents ~66 events per 1,000.
Statin Intolerance: The SAMSON Trial
The SAMSON (Self-Assessment Method for Statin Side-effects Or Nocebo) trial, published in NEJM 2020, used an elegant n-of-1 design to distinguish true pharmacological effects from nocebo phenomena.
60 patients who had previously stopped statins due to intolerable side effects entered a year-long protocol. Each month, they took either atorvastatin 20 mg, placebo, or nothing—in randomized order, blinded. They recorded daily symptom scores.
Results: On statin months, average symptom intensity was 8.0. On placebo months, it was 7.8. On no-tablet months, it was 6.7. The difference between statin and placebo was not statistically significant, but the difference between taking any tablet and taking nothing was. This proves that 90% of "statin side effects" are actually nocebo effects—caused by expectation of harm, not the drug itself.
After unblinding, 30 of 60 patients chose to restart statin therapy, suggesting that understanding the nocebo mechanism helped many overcome perceived intolerance.
PCSK9 Inhibitors: FOURIER and ODYSSEY OUTCOMES
PCSK9 inhibitors (evolocumab, alirocumab) represent a major advance in lipid-lowering therapy. These monoclonal antibodies prevent PCSK9 from degrading LDL receptors, dramatically increasing hepatic LDL clearance.
FOURIER (2017): 27,564 patients with established ASCVD on statin therapy were randomized to evolocumab or placebo. Evolocumab lowered LDL-C by 59% (from median 2.4 to 0.8 mmol/L). At median 2.2 years, cardiovascular events were reduced by 15% (HR 0.85). Importantly, ~500 patients achieved LDL-C <0.3 mmol/L with no excess adverse events.
ODYSSEY OUTCOMES (2018): 18,924 patients post-acute coronary syndrome randomized to alirocumab or placebo. Similar LDL reductions achieved. 15% reduction in major cardiovascular events. Notably, an exploratory analysis suggested mortality benefit (29% reduction) in the highest-risk tertile.
Cost-effectiveness: At initial list prices (~$14,000/year), PCSK9 inhibitors were not cost-effective for most patients. Price reductions (now ~$4,000-6,000/year) and restriction to high-risk patients have improved the value proposition.
Ezetimibe: IMPROVE-IT
Ezetimibe blocks intestinal cholesterol absorption via the NPC1L1 transporter. While it lowers LDL-C by only 15-20%, it does so through a mechanism independent of statins, making it an ideal add-on therapy.
The IMPROVE-IT trial (2015) randomized 18,144 post-ACS patients to simvastatin alone or simvastatin + ezetimibe. Adding ezetimibe lowered LDL-C from 1.8 to 1.4 mmol/L and reduced cardiovascular events by 6.4% over 7 years (NNT = 50). This was the first trial to demonstrate incremental benefit from LDL lowering below 1.8 mmol/L.
Bempedoic Acid: CLEAR Outcomes
Bempedoic acid inhibits ATP citrate lyase, an enzyme upstream of HMG-CoA reductase in the cholesterol synthesis pathway. Crucially, it requires activation by an enzyme (ACSVL1) present in liver but not skeletal muscle—explaining why it doesn't cause myalgia.
The CLEAR Outcomes trial (2023) randomized 13,970 statin-intolerant patients to bempedoic acid or placebo. LDL-C was reduced by ~20% (from 3.6 to 2.8 mmol/L). Major cardiovascular events were reduced by 13% (HR 0.87). This was landmark evidence that lowering LDL-C prevents events even in patients who cannot tolerate statins.
The "Lower is Better" Evidence
Multiple lines of evidence support the principle that lower LDL-C concentrations produce greater cardiovascular benefit:
- CTT meta-regression shows a strictly linear relationship between LDL reduction and event reduction down to the lowest achieved levels
- FOURIER patients achieving LDL <0.5 mmol/L had the lowest event rates with no safety concerns
- GLAGOV imaging study showed linear relationship between achieved LDL and plaque regression, with no floor effect
- Mendelian randomization studies show lifelong low LDL (PCSK9 loss-of-function) produces 3x the benefit of late-life statin therapy
Current guidelines (ESC/EAS 2019) recommend LDL-C targets of <1.4 mmol/L for very high-risk patients, with consideration of even lower targets in recurrent events. There is no evidence of harm from very low LDL-C levels in adults.
Special Populations
Elderly (>75 years): Evidence for primary prevention in the elderly is less robust. STAREE trial is ongoing. For secondary prevention (existing disease), benefit clearly extends to the elderly. Shared decision-making is appropriate.
Women: Women have historically been underrepresented in lipid trials. Available data suggest similar relative benefit but lower absolute benefit (due to lower baseline risk). Guidelines recommend equal treatment thresholds.
Diabetes: Diabetes is a "risk equivalent" in most guidelines. Even without established ASCVD, diabetic patients benefit from statin therapy. Moderate-to-high intensity statin is typically recommended.
Chronic kidney disease: Statins benefit CKD patients not on dialysis. For dialysis patients, evidence is mixed (SHARP showed benefit, but 4D and AURORA did not in diabetic dialysis patients).
References
- Cholesterol Treatment Trialists' Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670-1681. PubMed →
- Howard JP, Wood FA, Finegold JA, et al. Side Effect Patterns in a Crossover Trial of Statin, Placebo, and No Treatment. J Am Coll Cardiol. 2021;78(12):1210-1222. PubMed →
- Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease (FOURIER). N Engl J Med. 2017;376(18):1713-1722. PubMed →
- Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome (ODYSSEY OUTCOMES). N Engl J Med. 2018;379(22):2097-2107. PubMed →
- Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes (IMPROVE-IT). N Engl J Med. 2015;372(25):2387-2397. PubMed →
- Nissen SE, Lincoff AM, Brennan D, et al. Bempedoic Acid and Cardiovascular Outcomes in Statin-Intolerant Patients (CLEAR Outcomes). N Engl J Med. 2023;388(15):1353-1364. PubMed →
- Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias. Eur Heart J. 2020;41(1):111-188. PubMed →
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC Guideline on the Management of Blood Cholesterol. J Am Coll Cardiol. 2019;73(24):e285-e350. PubMed →
Make an Informed Decision
Work with your healthcare provider to understand your true risk level and match treatment intensity appropriately. The goal is preventing disease, not just changing numbers.